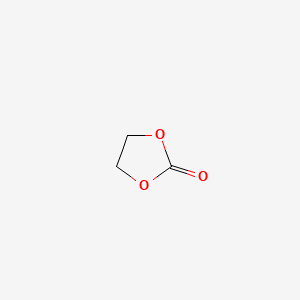


1,3-Dioxolan-2-one, 4-fluoro-
CAS No.: 114435-02-8
- Molecular Formula: C₃H₃FO₃
- Molecular Weight: 106.05 g/mol
Chemical type
- Fluorinated cyclic carbonate
- Electrolyte additive for lithium-ion batteries
Key properties
- Promotes formation of LiF-rich solid electrolyte interphase (SEI) on anode
- Enhances CEI formation on cathode when combined with Al(PO₃)₃
- Improves interfacial stability at high voltage and temperature
- Forms effective, robust SEI on SiOₓ-containing anodes
- Reduction potential of 0.2-0.3 V vs. Li/Li⁺
- LUMO energy of -0.7 eV
- Unstable in presence of transition metals, leading to decomposition and HF formation
- Prevents EC decomposition when used at sufficient concentration (e.g., 5 wt.%)
- Contributes to inorganic-rich SEI/CEI
- Anode SEI-forming additive
- Cathode CEI co-additive for synergistic protection
- Additive in LiPF₆/EC/EMC electrolytes for SiOₓ-Gr anodes
- Stabilizes SEI in high-voltage LIBs when paired with Sulfonyl diimidazole
- Suppresses lithium metal plating and capacity fade
- Employed at 5 wt.% in formulations for NMC811 | SiOₓ-Gr cells
Classification by use
- Electrolyte additives for lithium-ion batteries
- Chemicals used to suppress lithium dendrite growth
- SEI-forming additives
- Electrolyte components for silicon-based anodes
- Additives for high-voltage battery applications
A trustworthy factory and manufacturer
- [Cite:1] Aluminum metaphosphate-derived CEI suppresses interfacial degradation of NCM811 at 4.5 V and 60 °C, Electrochemistry Communications, Volume 184, March 2026, 108114
- [Cite:2] Sulfonyl diimidazole to stabilize fluoroEthyl methyl carbonate-based SEI in high-voltage Li ion cells with a SiOx containing negative electrode, Energy Storage Materials, Volume 72, September 2024, 103735