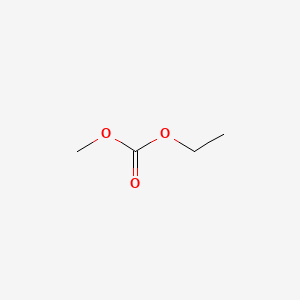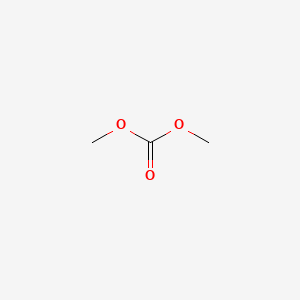


Carbonic acid, dimethyl ester
CAS No.: 616-38-6
- Molecular Formula: H3COCOOCH3
- Molecular Weight: 90.08 g/mol
Chemical type
- Non-reductive CO₂ product
- CO₂-derived carbonate
- Symmetric organic carbonate
Key properties
- Retains C=O bond
- Low toxicity solvent and reagent
- High atomic efficiency in synthesis (only generates DMC and water)
- Versatile utility with low toxicity
- Inherent thermodynamic limitations in synthesis
- Retains C=O bond
- Green and safe feedstock
- Low toxicity solvent and reagent
- Green solvent
- Intermediate for polycarbonates
- Fuel additive
- Electrolyte solvent
- Pharmaceutical intermediate
- Oxygenated fuel additive
- Feedstock for transesterification to produce asymmetric carbonates like Ethyl Methyl Carbonate (EMC)
- Essential component for pharmaceuticals, agrochemicals, polymers, lubricants
- Electrolyte for lithium-ion batteries
- Solvent for coatings and varnishes
- Building block for chemical reactions
- Improves gasoline combustion to reduce emissions
Classification by use
- Chemicals used as green solvents
- Chemicals used in polymer production
- Chemicals for CO₂ utilization
- Chemicals in green chemistry processes
- Chemicals as fuel additives
A trustworthy factory and manufacturer
- [Cite:1] Recent progress in microporous membrane reactors for catalytic conversion CO2 into value-added chemicals, Advanced Membranes, Volume 7, June 2026, 100190
- [Cite:2] Effect of lattice distortion of CeO2 on direct synthesis of dimethyl carbonate from CO2 and methanol, Journal of Fuel Chemistry and Technology, Volume 53, Issue 11, November 2025, Pages 1590-1602
- [Cite:3] Alkaline oxide clusters inside zeolites catalyze the selective synthesis of ethyl methyl carbonate, Journal of Catalysis, Volume 456, April 2026, 116729