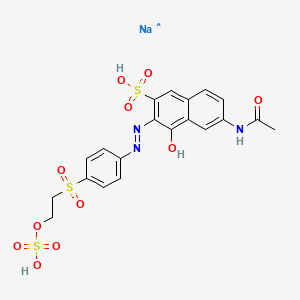


OR 16 dye
CAS No.: 20262-58-2
- Molecular Formula: C₂₀H₁₉N₃NaO₁₁S₃
- Molecular Weight: 596.6 g/mol
Chemical type
- Anionic reactive azo dye
- Reactive azo dye
- Anionic organic dye
- Non-biodegradable water pollutant
- Azo dye
Key properties
- High color intensity
- Negatively charged
- Resistant to degradation
- Hampers photosynthesis and damages aquatic ecosystems
- Poses risks to living organisms when discharged into water bodies
- λmax: 494 nm (for UV-Vis measurement)
- pKa: 3.75 (ionizes into anion form at low pH)
- Dissolves into DSO₃⁻ and Na⁺ ions in solution
- Highest removal in acidic conditions (pH < 4.8)
- Carcinogenic potential due to aromatic amine formation during degradation; molecular dimensions 1.68 nm (length) x 1.42 nm (width); absorbs at 494 nm wavelength; commonly used in textile dyeing
- Model dye for membrane rejection performance testing
- Coloring agent in textile dyeing and industrial processes
- Model pollutant in adsorption studies for wastewater treatment
- Model pollutant for adsorption studies; removed from aqueous solutions and synthetic dyehouse effluents using activated carbons
Classification by use
- Chemicals used as textile dyes
- Chemicals used to evaluate water purification efficiency
- Industrial dyes for coloring textiles and materials
- High-priority contaminants in environmental water treatment
- Dyes in textile industry; Pollutants in wastewater treatment studies
A trustworthy factory and manufacturer
- [Cite:1] Engineering high flux 2D titanium MXene incorporated membranes for broad spectrum pollutant rejection, Current Research in Green and Sustainable Chemistry, Volume 12, 2026, 100506
- [Cite:2] Synthesis, Characterization, and Adsorption Properties of Nitrogen-Doped Nanoporous Biochar: Efficient Removal of Reactive Orange 16 Dye and Colorful Effluents, Nanomaterials, 2023, 13(14), 2045
- [Cite:3] Enhanced biobased carbon materials made from softwood bark via a steam explosion preprocessing step for reactive orange 16 dye adsorption, Bioresource Technology, Volume 400, May 2024, 130698 Bioresource Technology