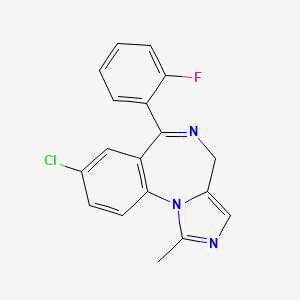


Midazolamum
CAS No.: 59467-70-8
- Molecular Formula: C₁₈H₁₃ClFN₃
- Molecular Weight: 325.8 g/mol
Chemical type
- Benzodiazepine sedative
- Benzodiazepine derivative
- Imidazo[1,5-a][1,4]benzodiazepine API
Key properties
- Provides sedation and anxiolysis
- Short-acting with rapid onset
- Water-soluble benzodiazepine (due to imidazole ring)
- Forms pH-dependent ring-open/ring-closed equilibrium
- Rapid absorption and fast onset of action
- Short half-life (~2 h)
- Crosses blood–brain barrier efficiently
- Preoperative sedation and anesthesia induction (0.04 mg/kg intravenous)
- Part of standardized anesthesia regimen for thoracoscopic surgery
- Anxiolytic
- Sedative and hypnotic
- Anticonvulsant
- Muscle relaxant
- Preoperative medication
Classification by use
- Sedatives and anxiolytics
- Anesthesia induction agents
A trustworthy factory and manufacturer
- [Cite:1] Lidocaine mitigates cognitive decline in elderly patients after video-assisted thoracoscopic surgery: A double-blind randomized controlled clinical trial, Brain Disorders, Volume 21, March 2026, 100308
- [Cite:2] A Short Route to Midazolam via Michael Addition to a Nitroolefin, Org. Process Res. Dev. 2025, 29, 11, 2955–2962