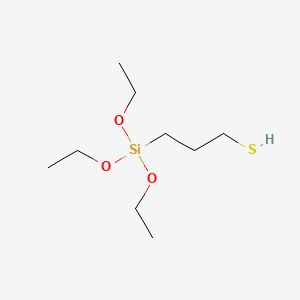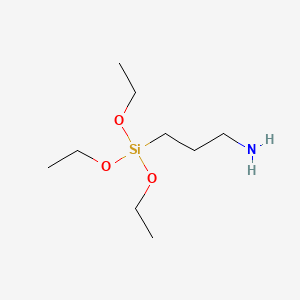


APTES
CAS No.: 919-30-2
- Molecular Formula: C₉H₂₃NO₃Si
- Molecular Weight: 221.37 g/mol
Chemical type
- Organosilane coupling agent
- Amino-functional silane
- Silane coupling agent
- Surface modifier
Key properties
- Contains amino (-NH2) and ethoxysilane groups for polymerization
- Controls nanosphere size in TAP NPs synthesis
- Forms Si-O-Si networks and -C=N bonds
- Concentration 0.6-2.1 mg/mL tunes properties
- Contains amino (–NH₂) groups
- Hydrolyzes to form Si–OH groups under acidic conditions
- Chemically grafts onto silica surfaces
- Forms stable Si–O–Fe bonds on magnetite surface; introduces hydrophilic –NH₂/–NH₃⁺ groups; enables covalent (imine) and electrostatic drug binding; confers biocompatibility and prevents agglomeration/oxidation of bare MNPs
- Reactant with TA to form TAP NPs in ethanol-Tris-HCl solution
- Provides reactive sites for hydrogen bonding with F127-F
- Adjusts physical topology for membrane roughness and superoleophobicity
- Surface modifier for silica aerogels to enhance dispersion in polymer matrices
- Improves interfacial adhesion via hydrogen bonding
- Surface functionalisation of magnetite nanoparticles to create a stable, biocompatible nanocarrier (APTES@MNPs) that supports high drug loading (up to 99 % at pH ~10) and controlled release
Classification by use
- Chemicals used in nanomaterial synthesis
- Chemicals for crosslinking and surface engineering
- Chemicals used as coupling agents
- Chemicals used in surface modification
A trustworthy factory and manufacturer
- [Cite:1] Construction of dual-heterogeneous membrane surface via nanosphere-mediated surface segregation for oil-water separation, Advanced Membranes, Volume 7, June 2026, 100205
- [Cite:2] Preparation and Performance of Thermal Insulation Waterborne Polyurethane Coatings Containing 3-Aminopropyltriethoxysilane Modified Organo-Bridged Silica Aerogel, Coatings, 2025, 15(9), 1021
- [Cite:3] APTES modified magnetite nanoparticles as a theranostic nanocarrier: a study of loading and sustained release of daunorubicin, Mater. Adv., 2025,6, 6843-6855